Weathering
Weathering is the process by which materials (both man-made and natural rocks, minerals, soils, and plants) are disintegrated or decomposed through contact with Earth’s geological subsystems. When weathering occurs, substances are broken down into smaller pieces in situ (in place) and therefore weathering should be distinguished from erosion, which necessitates movement of materials. Weathering is generally considered to fall under two classifications: physical (mechanical) weathering and chemical weathering.
Physical and chemical weathering processes do not act in isolation and most often act in conjunction. Weathering significantly alters our planet’s surface, helping to shape and define cliffs, caves, and beaches and influencing mineral structure and chemical composition.
Contents
Physical Weathering
Physical weathering is the breakdown of materials by mechanical forces. Put simply, physical weathering is the breakdown of rock or other materials into smaller pieces without chemical change. Physical weathering is the result of various processes, though the primary agent for physical weathering is abrasion, the mechanical scraping of rocks and minerals, which can occur by direct contact with atmospheric forces such as water, ice, and wind.
 Chemical weathering of iron via oxidation
Chemical weathering of iron via oxidation
Types of physical weathering include (but are not limited to) the following:
- frost (ice) wedging
- salt wedging
- exfoliation
- biological weathering
- root wedging
- animal burrowing.
Frost Wedging
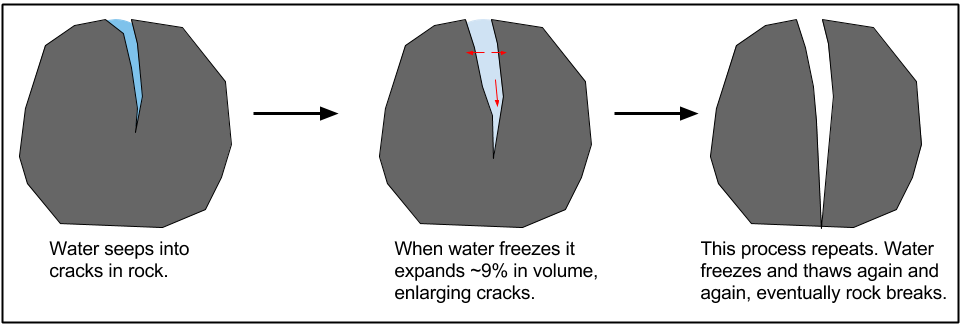 A diagram displaying the steps of frost wedging
A diagram displaying the steps of frost wedging
One type of physical weathering is frost wedging. Frost wedging occurs when liquid water seeps into pores and small cracks in the earth and expands when it freezes. This causes the cracks to enlarge. Cycles of freezing and thawing repeat and eventually result in the breaking of materials into smaller pieces.
Chemical Weathering
Chemical weathering is the breakdown of rocks via chemical reactions. Consequently, chemical weathering alters the composition of rocks. Water is frequently involved in the reactions that result in chemical weathering.
Types of chemical weathering include (but are not limited to) the following:
 Top equation: Formation of carbonic acid in acid rain. Bottom equation: Dissolution of calcium carbonate.
Top equation: Formation of carbonic acid in acid rain. Bottom equation: Dissolution of calcium carbonate.
- oxidation
- hydrolysis
- dissolution (usually via carbonation)
- hydration
- dehydration
- biological weathering via plant acids.
Carbonation
 Limestone karst in Tsingy de Bemaraha National Park, Madagascar
Limestone karst in Tsingy de Bemaraha National Park, Madagascar
Chemical weathering by carbonation usually is the result of acid rain. Acid rain is formed when carbon dioxide combines with water to form carbonic acid. When rain containing carbonic acid makes contact with rocks containing calcium carbonate \((\ce{CaCO3}),\) most often limestone, the acid dissolves it. The dissolution of limestone can lead to the formation of spectacular, unique geological features known as karsts (solutional caves).